SANDSTONE DIAGNOSTICS

Medical device prototyping, clinical validation, patent development. FDA approval success.
Sandstone is where I learned disciplined, end-to-end physical product development.
At a medical device startup, I volunteered for every learning opportunity. My role spanned R&D, manufacturing engineering, supply chain, operations, quality systems, regulatory affairs, FDA submissions, and patent writing. I wanted to understand how physical products actually get built and shipped in regulated industries.

Projects and Responsibilities
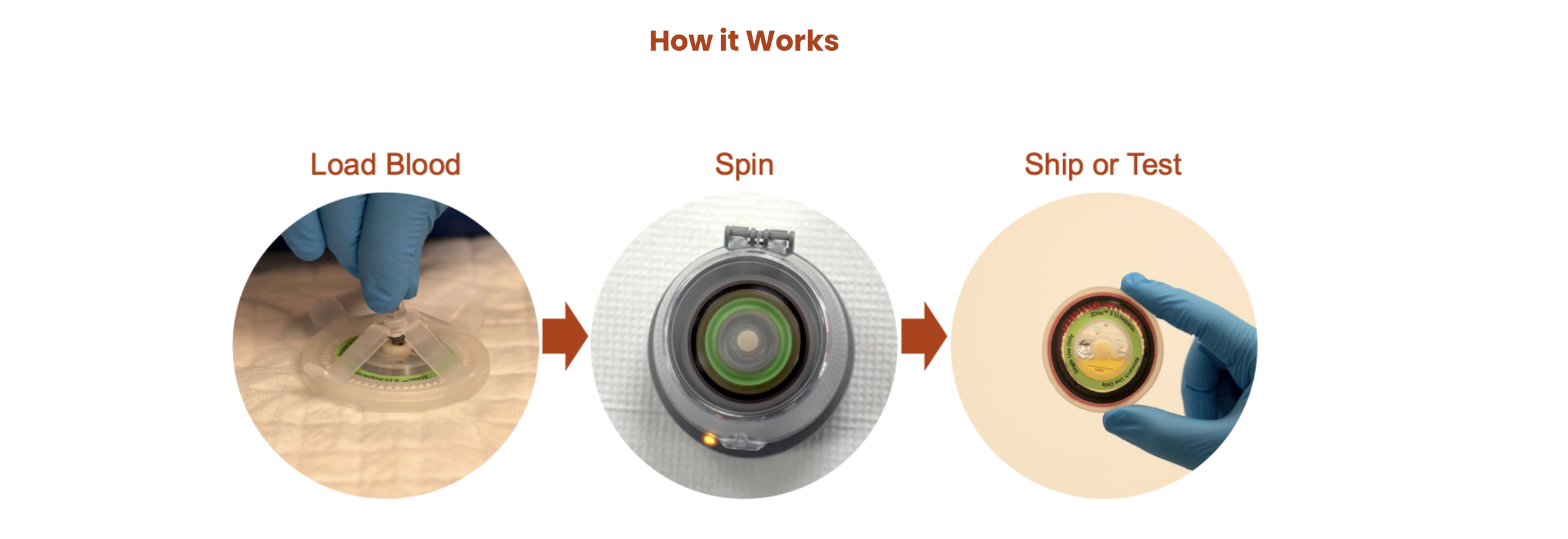
Torq Zero Delay Centrifuge. Portable, battery-operated device that fits in the palm of your hand. Stabilizes plasma/serum at point of collection.
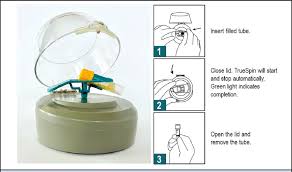
Labcorp Truespin. Medical device I conceptualized and developed.
Product Development
- • Prototyped medical devices (3D printing, soldering, assembly)
- • Designed manufacturing processes (injection molding tooling)
Intellectual Property
- • Patent writing and development
Research & Clinical
- • Coordinated clinical trials
- • Ran lab experiments and performance validation
Manufacturing & Supply Chain
- • Sourced and contracted suppliers
- • First article inspections
- • Bill of materials management
- • Package design and medical device labeling (standardized symbols, regulatory compliance)
Quality & Regulatory
- • Owned QA/QMS systems
- • FDA submissions and GUDID registration
- • Package verification testing
Results
Patents
- • Thermal Management (Granted)
- • Portable Centrifugation (Pending)
- • Centrifugal Tube Assembly (Closed)
- • Compact Design (Closed)
Company Outcome: Acquired by Labcorp
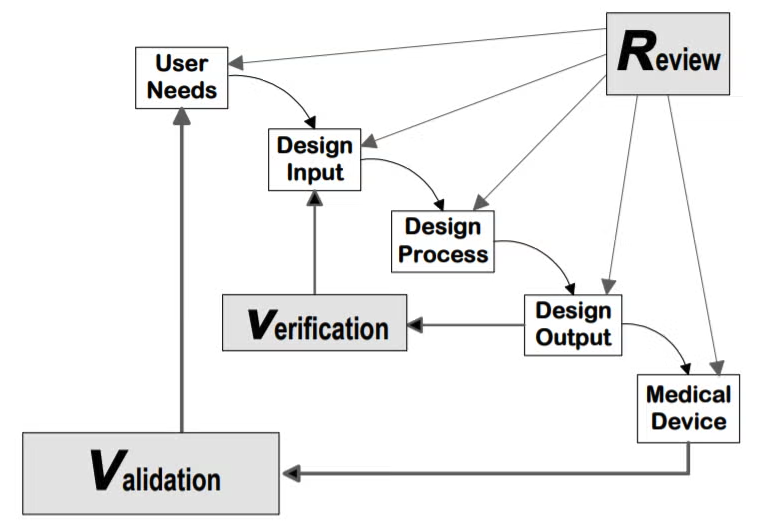
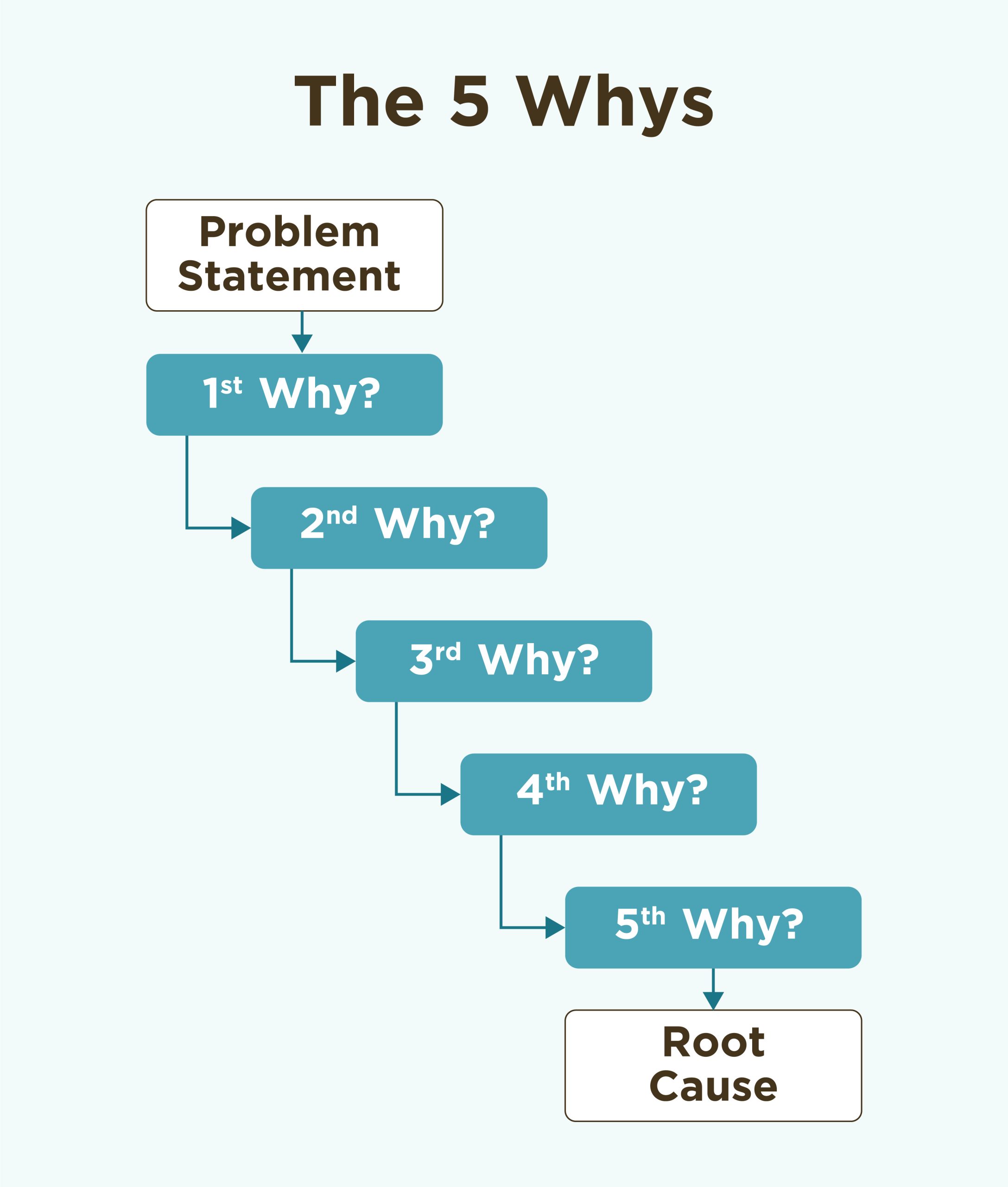
This experience taught me end-to-end product development in a rigorous environment. I learned how to operate fast and apply frameworks that transferred directly to software product work at scale.
Frameworks That Shaped How I Think
These frameworks that I learned at Sandstone became foundational to how I approach product work.